Gholson Lyon is a physician-scientist currently working at the Utah Foundation for Biomedical Research and the Center for Applied Genomics at Children’s Hospital of Philadelphia. He will be starting as an assistant professor in human genetics at Cold Spring Harbor Laboratory next month. I asked him to write this guest post to provide some personal context to his thought-provoking commentary in Nature (subscription required) on returning genetic findings to research subjects. [DM]
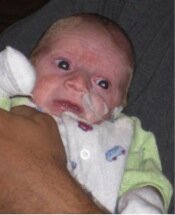
Photo of Max, who died aged four months from Ogden syndrome. Posted with permission from his family.
I have just published in Nature a commentary discussing the need to bring exome and genome sequencing into the clinical arena, so that these data are generated with the same rigorous clinical standards as for any other clinical test. This way, we can then easily return at least medically actionable results to research participants. In this day and age of consumer and patient empowerment, I can also see eventually returning all data, including the raw data, to any interested participants, as this can then promote crowd-sourcing for data analysis, with research participants controlling and promoting the relative privacy of and analysis of their own data.
As I described in my commentary, my thinking on this matter was prompted mainly by Max (see picture) and his family. The obituary for Max can be found here, and that of his cousin, Sutter, here. We described their condition here, and we named this new disease Ogden Syndrome in honor of where the first family lives. I am now trying to think about and discuss the human aspects of and lessons from this story. My thinking has also been influenced somewhat by the late James Neel, who wrote a very thought-provoking book called Physician to the Gene Pool.
To me, it was deeply disconcerting that I could not officially return any results to this family (or to another family in a different project discussed here) even when the papers describing the genetic basis of their disease were published, as this was considered “research” and was not performed in a clinically appropriate (CLIA-certified) manner. This was all the more painful when one of the sisters in the Ogden family became pregnant and asked me what I knew. I cannot predict whether it would have helped or hurt this woman to learn during her pregnancy that she was indeed a carrier of the mutation, with the associated 50% risk of her baby boy having the disease. I also do not know if she would have undergone any genetic testing via amniocentesis of the fetus prior to birth (with the associated ~1% risk of miscarriage from the procedure), nor do I know what decisions she might have made prior to the birth even if she had undergone such testing. All in all, it was certainly an ethical and moral dilemma for me not to be able to return the research result to her, given that the results were not obtained in a CLIA-certified manner. It is still an issue, as there are even now financial and systematic barriers for getting all women in the family tested with a CLIA-certified gene test for NAA10 (which was developed over a six month period by ARUP Laboratories). It would have been so much better if we had just done the entire sequencing up front in a CLIA-certified manner.
Continue reading ‘Guest post: Time to bring human genome sequencing into the clinic’
 RSS
RSS Twitter
Twitter
Recent Comments